Never start from scratch
Stop copying from old documents and fixing outdated content. Start with a guided form pre-loaded with the right options.

What if you could write any URS in 15 minutes?
Transform your approved Word templates into guided forms. Generate consistent, audit-ready GMP documents in minutes.
Be first to know when we launch. No spam, ever.

Fill a guided form, generate a complete document
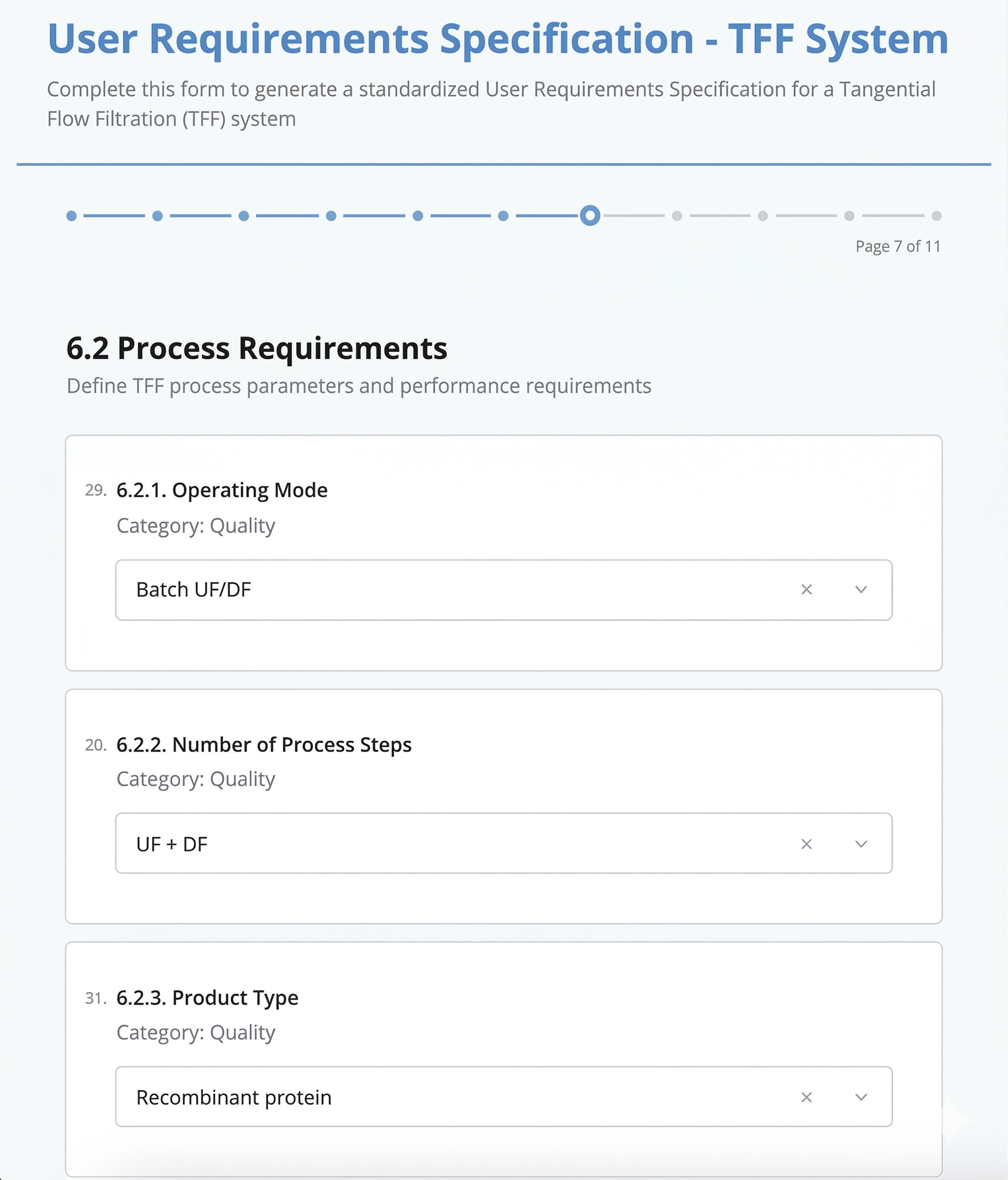
Guided form with smart dropdowns
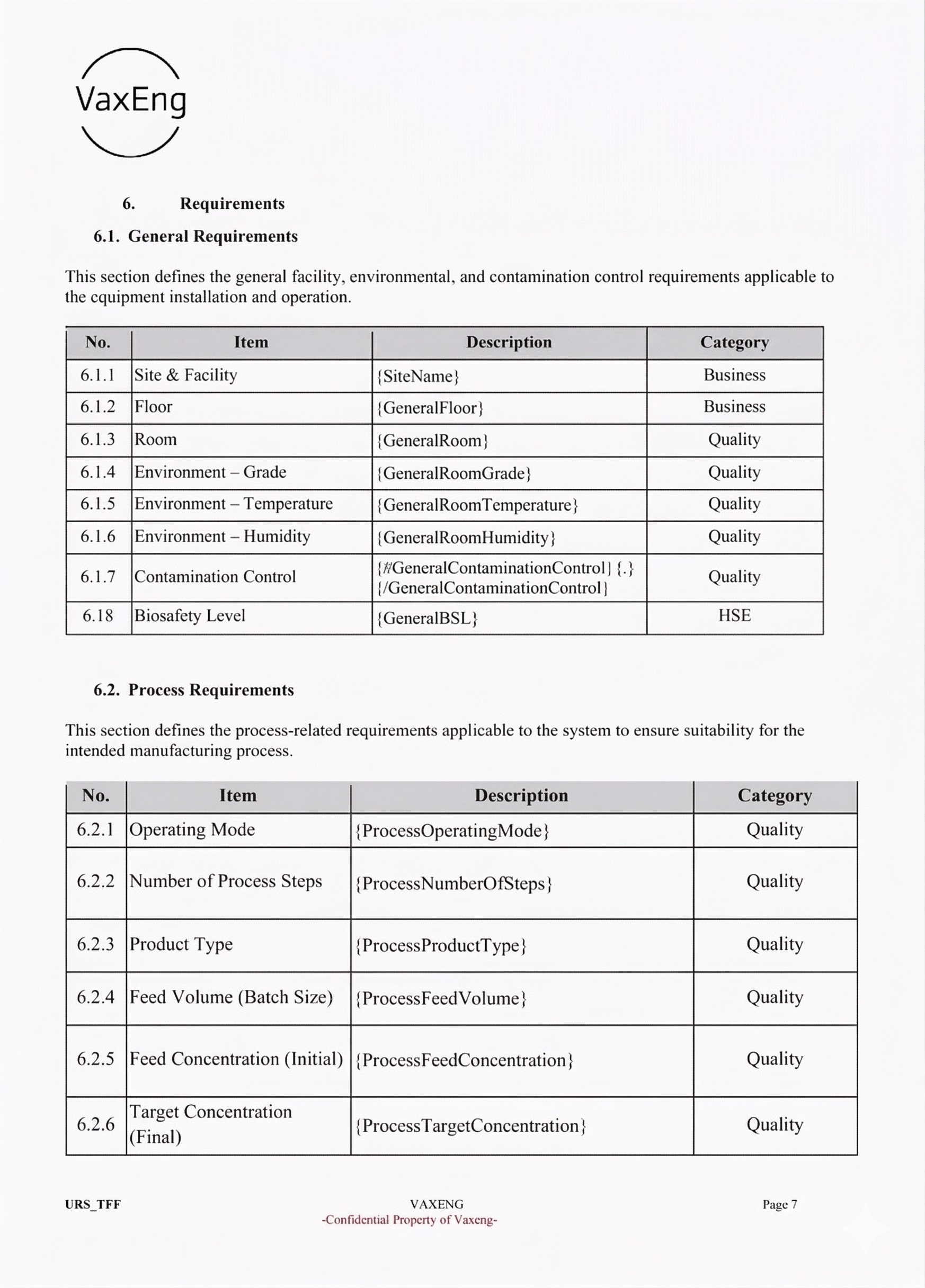
Generated Word document
Stop copying from old documents and fixing outdated content. Start with a guided form pre-loaded with the right options.
Tables, headers, numbering — all handled automatically. Your output matches the approved template every time.
Guided forms ensure every parameter is considered. No more forgotten specifications or incomplete sections.
EnjiPro works with standard biopharma document structures. Start with our pre-built templates or import your company's approved formats.
Covering the full spectrum of biopharma manufacturing documentation

EnjiPro is designed with pharmaceutical manufacturing requirements in mind. Ensure traceability, consistency, and regulatory compliance across all your documentation.

Founder — Biopharmaceutical Engineer & CMC Consultant
10+ years in vaccine and biologic manufacturing. 15+ vaccine programs across 13 countries. 4 GMP facility constructions led. Contributed to two approved COVID-19 vaccines. After years of writing URS, equipment specs, and validation documents by hand, I built EnjiPro to give engineering teams their time back.
Join the waitlist and be among the first to experience EnjiPro.